Chapter 3
Fighting an Invisible Enemy
Humans have evolved along with bacteria and viruses for millions of years, so it should be no surprise that the human body has developed a system of keeping the harmful microbes out. The first line of defense is the skin, which is a sheath of closely interlocking cells. Sweat and oil glands under the skin secrete acids that prevent the growth of microbes, and harmless bacteria that normally live on the body defend their territories against foreign bacteria.
The nose, mouth, and throat are common sites of attack, but fragile membranes and layers of sticky mucus that are toxic to harmful microbes protect them. Tears and saliva also contain antiseptic substances. Bacteria and viruses are sneezed out, coughed up, and cried out of the body. If microbes make it through these barriers, they are swallowed. Most will not survive in the stomach's acid environment or the toxic world of the intestines. The beneficial bacteria that reside there will fight off potential competitors.
Our Internal Army
A cut on the skin is a way in for some opportunistic microbes, but the body quickly fights back with an inflammatory response. Injured cells release histamine, a chemical that causes blood vessels near a wound to swell, which brings more blood to the area to aid healing. Blood quickly clots to seal off the cut and prevent more bacteria from getting into the bloodstream and spreading further into the body.
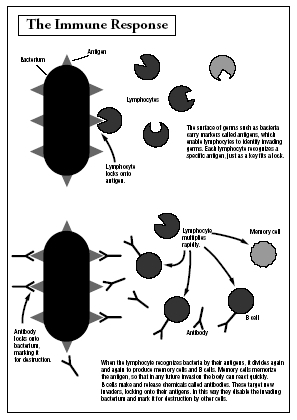
Injured cells also release a chemical that attracts bacteria-eating white cells called phagocytes. Phagocytes engulf any bacteria they encounter until they are so full they die. Any bacteria that manage to get past the phagocytes will be carried to other parts of the body. The body's immune system is alerted and sends out lymphocytes, or white cells. There are about 2 trillion lymphocytes patrolling a person's bloodstream and lymph system at any given time. They stand guard in the spleen, tonsils, and lymph nodes and detect all foreign invaders such as bacteria, viruses, fungi, and transplanted organs.
When a foreign invader is detected, the lymphocytes divide and create antibodies to kill or neutralize the invaders. Antibodies are chemicals that target and destroy only one type of invader. For example, an antibody sent out to fight against the pneumonia bacterium cannot fight a salmonella bacterium. It takes about a week to generate enough antibodies to fight a disease. In that time, it is a race for survival between man and microbe.
The immune system has an ingenious way of remembering past battles so that this life-and-death struggle does not happen again. Lymphocytes create memory cells that circulate through the body ready to battle the old enemy. If a person comes down with the same strain of pneumonia a second time, the memory cells kick into maximum production immediately. Some memory cells last a lifetime, which provides immunity against that disease for the rest of a person's life. That is why a person who has had chicken pox will not get it again. This amazing immune response is the principle behind vaccinations.
Ancient Asian Secrets
Smallpox ravaged Asia for generations, yet as early as the eleventh century there was a method of fighting
smallpox called ingrafting. No one knows how it was developed, but over time, a treatment was devised where pus from the sore of a person with a mild case of smallpox was smeared into a scratch on the arm of a healthy person. The person who was ingrafted usually developed mild symptoms but recovered quickly. They never caught smallpox again.
This technique spread to Europe with the help of Lady Mary Wortley Montagu, the wife of the British ambassador living in Turkey in 1716. After witnessing this procedure, she had her three-year-old son ingrafted. She brought news of the technique back to England, where it was called inoculation. The procedure was not widely used because, although it was successful, there was a risk that the patient could develop a severe case of the disease.
The First Vaccine
One boy who was inoculated was eight-year-old Edward Jenner. He survived the painful procedure and grew up to be a country doctor whose keen observations led to the first vaccinations.
In 1796 Jenner noticed that young women who milked cows sometimes became infected with cowpox, a disease that caused sores on a cow's udders. The girls would get painful sores on their hands, but the disease was not fatal. Once the girls caught cowpox, they never became infected with smallpox.
In May of that year Jenner performed his first cowpox experiment. He took fluid from a sore on the hand of dairymaid Sarah Nelmes and inoculated a healthy eight-year-old boy named James Phipps. Within a few days James came down with a fever and a small sore. On July 1, believing that the cowpox inoculation would prevent the development of smallpox, Jenner inoculated James with matter from a smallpox patient. Nineteen days later Jenner wrote, "The Boy has since been inoculated for the Smallpox which as I ventured to predict produced no effects. I shall now pursue my Experiments with redoubled ardor." 15
Jenner went on to repeat his experiments and published his results, calling his technique vaccination and the matter taken from the cowpox sore a vaccine (derived from vacca , the Latin word for cow). In a letter written to a friend, Jenner predicted, "The annihilation of smallpox—the most dreadful scourge of the human race—will be the final result of this practice." 16 Jenner would never know how accurate his prediction would become. He also would never know how his vaccine worked or even what kind of organism he was actually fighting against. That information would not come for many more years.
How Vaccines Work
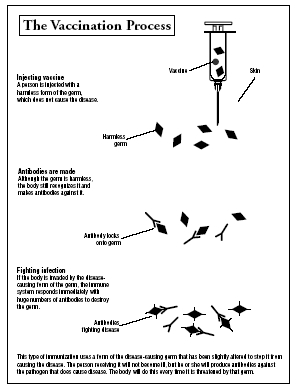
Vaccines work by using our own natural defenses. An injection of a weakened virus or a part of a virus is just enough to trigger the lymphocytes to create memory cells. If the virus invades again, the body is able to fight it with ready-made antibodies before a serious infection takes hold. Vaccines may be made with dead microbes or parts of dead viruses. Some are made with living microbes that are weakened and rendered harmless but are still able to elicit an immune response.
Rabies Vaccine
Louis Pasteur had created a successful vaccine for chickens, another for sheep, and he had been experimenting on a rabies vaccine for dogs, but he had not developed a safe vaccine for humans. But that did not matter to the mother of nine-year-old Joseph Meister, who took her son to Pasteur's office in 1885. A mad dog had bitten Joseph. Rabies is a horrible disease that infects only mammals. The virus attacks the nervous system and infects the brain, causing a difficult and painful death.
Pasteur knew that a weakened germ worked as a vaccine against other diseases in animals and believed that a similar treatment for humans should work against rabies. He injected Joseph with the weakened vaccine and increased the dose daily. After fourteen days Joseph Meister was stronger and had made history. He became the first person to survive rabies.
More Vaccines
Pasteur's success inspired a concerted effort to develop vaccines for other dreadful diseases, but it did not happen quickly. Max Theiler created a vaccine against yellow
fever in the 1920s, and Jonas Salk and Albert Sabin produced polio vaccines in the 1950s.
Today, there are vaccines for mumps, rubella, measles, tetanus, chicken pox, flu, and other once dangerous diseases. But there are many viral infections that the medical community cannot prevent with an injection. For example, there are too many strains of the common cold to create an effective vaccine, and other viruses mutate too quickly. A virus can alter its outer protein coat so that antibodies and vaccines that once worked on the virus are no longer effective. Even a slight difference can mean that a person's memory cells would not recognize the virus, and the antibodies that a person's immune system created in the past would be powerless against this new strain.
The First Antibiotics
It seems ironic that medical researchers were successful in developing a preventive medicine for viruses, which were unknown to science and unseen by man, but were not able to fight off known bacterial infections. The key was finding a way to kill a bacterium cell without harming human cells.
The first breakthrough came in 1910, when Paul Ehrlich, a German scientist, discovered that an arsenic compound killed a spiral-shaped bacterium called a spirochete that caused syphilis. His discovery was inspired by Robert Koch's work with aniline dye. In a meeting in Berlin, Ehrlich heard Koch describe how he used the dye to identify the tuberculosis bacterium. When Koch applied the dye, it stained the bacteria cells, making them easier to see under a microscope, but it also killed the microbes.
Ehrlich experimented with many other dyes and chemical compounds before he achieved success with arsenic compound 606, which was dubbed "the magic bullet."
How Antibiotics Work
Ehrlich's compound was called a magic bullet because, like other antibiotic agents, it specifically targeted the syphilis spirochete. Antibiotics are simply chemicals that react with other chemicals. Every cell, whether it is human or bacterium, is also made up of chemicals. The cell's membranes are covered with receptor sites that allow the cell to react with or take in other chemicals. In order for an antibiotic to work, it must have the right chemical makeup, or key, to fit the chemical makeup, or lock, at the receptor site on the bacterium. But the antibiotic's chemical key must not fit the receptor sites of other cells in the patient's body. If it does, then it would cause adverse side effects.
Each kind of antibiotic attacks a bacterium in a different way. Some, like penicillin, stop the bacteria from forming a cell wall. Other antibiotics interfere with the bacteria's ability to make essential nutrients, such as folic acid and other proteins, while others stop the bacteria's DNA replication.
Penicillin
The power of microorganisms can be harnessed to heal as well as harm. This was discovered by chance when Alexander Fleming observed a yellowish mold growing on a bacterial culture in his lab. It was 1928, and Fleming had been studying staphylococcus, which is a common bacterium on skin. He noticed that wherever the mold grew, an area of clear liquid surrounding it was free of bacteria. Wherever Fleming spread the mold juice, which Fleming called penicillin, bacterial growth was stopped dead. Although penicillin worked wonders, Fleming was unable to present it to the public. Apparently people were not ready to accept a microorganism that could make an effective antibiotic.
Twelve years later, a young Australian doctor named Howard Walter Florey, along with his colleagues Ernst Boris Chain and Dr. Norman G. Heatley, showed the world that penicillin was indeed a miracle drug. It killed the bacteria that caused scarlet fever, pneumonia, diphtheria, and meningitis, as well as other common bacterial infections.
The discovery of penicillin was so important that American soldiers were sent to collect soil samples from India, China, Africa, and South America so that it could be tested for other miracle molds. Employees of the U.S. Department of Agriculture laboratory in Peoria, Illinois, were instructed to collect any unusual molds as well. One employee, Mary Hunt, earned the nickname Moldy Mary because she searched through people's garbage cans and litter. While poking through a neighbor's trash, Mary found a rotting cantaloupe that had a golden mold growing on it. When the melon mold was tested, it produced twice as much penicillin as Fleming's mold, and it grew easily in large quantities. It was named Penicillium chrysogenum , and it replaced Fleming's mold for use in penicillin production until researchers learned to make the drug synthetically.
Drugs Dug from the Earth
Every pharmaceutical company raced to find new antibiotic compounds. Bristol-Meyers Pharmaceuticals

sent envelopes to all of its stockholders with instructions to collect soil samples from their neighborhoods. Other companies contacted missionaries in far-off places, foreign news correspondents, airline pilots, and deep-sea divers in their search for a mold that might lead to a new treatment.
In 1943 Dr. Paul Burkholder at Yale University sent out plastic mailing tubes to everyone he knew and received more than seven thousand soil samples in return. One soil sample sent from Venezuela contained a powerful antibiotic that was eventually developed into the drug called Chloromycetin. It killed many different kinds of microbes, including the deadly bacteria that caused Rocky Mountain spotted fever and typhus.
Dr. Selman Waksman at Rutgers University worked for the pharmaceutical firm Merck & Company. He tested the mold found in the throat of a New Jersey chicken. It contained a compound called streptomycin that killed the tuberculosis bacillus—something penicillin could not do.
Man over Microbes
With the development of antibiotics, common infectious diseases lost their grip on the world. People believed that man had conquered harmful bacteria and viruses. The medical community even fulfilled Jenner's prediction of annihilating smallpox.
By the end of the 1800s, many European countries had enacted laws requiring its citizens to be vaccinated against smallpox. In short order, the virus disappeared from many countries. Surprisingly one of the last Western countries to eradicate smallpox from within its borders was the United States, in the late 1940s.
The success of mandatory vaccination inspired officials at the United Nations to adopt a resolution to eradicate smallpox from the forty-four countries that still reported its occurrence. The World Health Organization (WHO), which is part of the United Nations, set a deadline of January 1, 1977.
Teams of medical workers searched for outbreaks of smallpox in poor pockets of major cities and remote villages. Wherever an outbreak occurred, the team swooped in to vaccinate all the inhabitants, creating a ring of containment around the victims in a particular area. Those who were infected were put into quarantine. Defusing each epidemic case by case and country by country, the WHO successfully snuffed out the once raging flames of smallpox.
By 1979 the WHO declared the project a success. The only places on Earth where smallpox existed were in laboratory test tubes. One question remained: What should be done with the stored virus? Some nations voluntarily destroyed their supplies, and others handed them over to research centers in the Soviet Union or the United States. There are now about four hundred vials of frozen virus securely stored at the U.S. Centers for Disease Control in Atlanta, Georgia, and another two hundred stored in a lab in Moscow. Although the WHO ordered the destruction of all smallpox samples in 1993, the order was not carried out. Scientists still debate the validity of destroying a virus species. Some argue that more study could provide clues to fighting other deadly microbes.
Fighting Back
The elimination of smallpox and the availability of so many antibiotics lulled the world into believing that infectious diseases were a thing of the past. The U.S. surgeon general William H. Steward even declared before Congress in 1969 that he was ready to "close the book" 17 on infectious disease. The development of antibiotics was put on the back burner.
But as quickly as man could manufacture antibiotics and vaccines, bacteria and viruses were faster to develop resistance to the drugs. In an interview with Newsweek , Dr. Richard Wenzel of the University of Iowa said, "Ever since 1928, when Alexander Fleming discovered penicillin, man and microbe have been in a footrace. Right now the microorganisms are winning. They're so much older than we are … and wiser." 18
Microbes Mutate
The wisdom of a microbe lies in its ability to change. They are able to reproduce much faster than their human competitors. A new generation can come along as quickly as every fifteen minutes, and each time a bacterium divides, there is a chance for error. A random change in the genetic makeup of a cell that becomes a permanent inherited characteristic is called a mutation. And a mutation that increases a microbe's chance of survival is passed on to the next generation.
Bacteria can also trade or share parts of their DNA through a process called horizontal gene transfer. In addition to strands of DNA, bacteria have rings of DNA called plasmids. These plasmids give the bacteria certain survival skills, such as being resistant to a type of antibiotic. This means that bacteria in the same generation can potentially share advantageous plasmids, just as easily as two friends exchange phone numbers. Two bacteria can exchange a gene or genes that allow them to inhabit a new species of animal, thrive in a new climate, or protect them against a certain drug.
Growing Resistance
The use of antibiotics creates a situation in which the fittest microbes survive. Each time someone uses an antibiotic, the majority of the bacteria are killed but not all. These heartier microbes are left to multiply and spread their resistance to the next generation. Within four years of the widespread use of penicillin in the 1940s, doctors saw evidence of microbes that had grown resistant to it. Even Alexander Fleming himself warned the public in a New York Times interview of the dangers of taking antibiotics. "The microbes are educated to resist penicillin and a host of penicillin-fast [resistant] organisms is bred out which can be passed to other individuals and from them to others until they reach someone who gets a septicemia or a pneumonia which penicillin cannot save." 19
As each new antibiotic came on the market, a microbe came along that could withstand the toxic effects. Today doctors are encouraged to stop prescribing antibiotics for viral infections, because they have no power over a virus. And when patients are prescribed an antibiotic, they need to take the entire dose. After two days on an antibiotic, a patient usually starts to feel better, but that is only because the antibiotic has killed off a significant number of bacteria. The minute treatment is stopped, the surviving bacteria begin to multiply. It takes only one drug-resistant bacterium to multiply into millions.
The Bacteria Eaters
One treatment that may prove to be a solution to the problem of antibiotic resistance comes from an unlikely source—viruses. It is a method developed in Russia before Fleming discovered penicillin. To modern mentality it seems bizarre, for it pits microbes against each other.
Phage therapy harnesses specific kinds of viruses that attack only certain harmful bacteria. Discovered and named by Felix d'Herelle in 1917, bacteriophages (bacteria-eating viruses) were soon used by doctors to
cure cholera and typhoid fever. Although the treatment never caught on in the West, research continues, particularly in the Republic of Georgia. Today the world's foremost center for the development of phage therapy is at the Eliava Institute in Tbilisi, Georgia. Doctors at the institute study a wide range of viruses collected from nature. The viruses that are active against harmful bacteria are then cultivated and used for treatment. A patient suffering from an antibiotic-resistant bacterial infection is injected with a solution that contains the proper bacteriophages. The viruses seek out the larger harmful bacteria and inject their genetic material into the cell, where it hijacks the bacteria's reproductive machinery. Only one hundred bacteriophages placed on an infected wound is enough to destroy more than 100 million bacteria. Once the bacteria are eliminated, the viruses also die out. They have no cells to infect and are washed harmlessly out of the patient's body.
More than twenty companies in the United States are now studying and testing phage therapy in the lab and will seek future government approval to conduct clinical trials on humans. In the meantime, dozens of exotic, mysterious illnesses are cropping up all over the world.
-Alexa Horn
I am an eight grade student and is currently working on a project. I would like to ask permission to use one of your photos which would be the nineteenth century painting of the black death of 1340.
I will only use it for this project and will cite it properly. By the way, the information on your website are very educational and awesome. Thank you. Jasmine
I'm a secondry school student and i am working on a project involving vaccines and bactera, I would like to use two pictures involuing how the vaccine and bacteria work and i will cite it properly useing harvard style bibliography. this site is also very helpful in finding the information I needed thanks.