Nitrogen cycle
The term nitrogen cycle refers to a series of reactions in which the element nitrogen and its compounds pass continuously through Earth's atmosphere, lithosphere (crust), and hydrosphere (water component). The major components of the nitrogen cycle are shown in the accompanying figure. In this diagram, elemental nitrogen is represented by the formula N 2 , indicating that each molecule of nitrogen consists of two nitrogen atoms. In this form, nitrogen is more correctly called dinitrogen.
Nitrogen fixation
Nitrogen is the most abundant single gas in Earth's atmosphere. It makes up about 80 percent of the atmosphere. This fact is important because plants require nitrogen for their growth and, in turn, animals depend on plants for their survival. The problem is, however, that plants are unable to use nitrogen in its elemental form—as dinitrogen. Any process by which elemental dinitrogen is converted to a compound is known as nitrogen fixation.
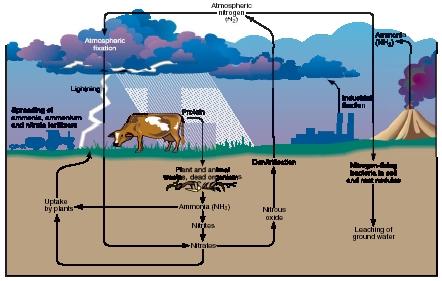
Dinitrogen is converted from an element to a compound by a number of naturally occurring processes. When lightning passes through the atmosphere, it prompts a reaction between nitrogen and oxygen; oxides of nitrogen—primarily nitric oxide (NO) and nitrogen dioxide (NO 2 )—are formed. Both oxides then combine with water vapor in the atmosphere to form nitric acid (HNO 3 ). Nitric acid is carried to the ground in rain and snow, where it is converted to nitrites and nitrates. Nitrites and nitrates are both compounds of nitrogen and oxygen, the latter containing more oxygen than the former. Naturally occurring minerals such as saltpeter (potassium nitrate; KNO 3 ) and Chile saltpeter (sodium nitrate; NaNO 3 ) are the most common nitrates found in Earth's crust.
Certain types of bacteria also have the ability to convert elemental dinitrogen to nitrates. Probably the best known of these bacteria are the rhizobium, which live in nodules on the roots of leguminous plants such as peas, beans, clover, and the soya plant.
Finally, dinitrogen is now converted to nitrates on very large scales by human processes. In the Haber process, for example, nitrogen and hydrogen are combined to form ammonia, which is then used in the manufacture of synthetic fertilizers, most of which contain nitrates.
Words to Know
Ammonification: The conversion of nitrogen compounds from plants and animals to ammonia and ammonium; this conversion occurs in soil or water and is carried out by bacteria.
Denitrification: The conversion of nitrates to dinitrogen (or nitrous oxide) by bacteria.
Dinitrogen fixation (nitrogen fixation): The conversion of elemental dinitrogen (N2) in the atmosphere to a compound of nitrogen deposited on Earth's surface.
Nitrification: The process by which bacteria oxidize ammonia and ammonium compounds to nitrites and nitrates.
Ammonification, nitrification, and denitrification
Nitrogen that has been fixed by one of the mechanisms described above can then be taken in by plants through their roots and used to build new stems, leaves, flowers, and other structures. Almost all animals obtain the nitrogen they require, in turn, by eating plants and taking in the plant's organic forms of nitrogen.
The nitrogen stored in plants and animals is eventually returned to Earth by one of two processes: elimination (in the case of animals) or death (in the case of both animals and plants). In whatever form the nitrogen occurs in the dead plant or animal, it is eventually converted to ammonia (NH 3 ) or one of its compounds. Compounds formed from ammonia are known as ammonium compounds. This process of ammonification is carried out (as the plant or animal decays) by a number of different microorganisms that occur naturally in the soil.
Ammonia and ammonium compounds, in their turn, are then converted to yet another form, first to nitrites and then to nitrates. The transformation of ammonia and ammonium to nitrite and nitrate is an oxidation process that takes place through the action of various bacteria such as those in the genus Nitrosomonas and Nitrobacter. The conversion of ammonia and ammonium compounds to nitrites and nitrates is called nitrification.
In the final stage of the nitrogen cycle, oxygen is removed from nitrates by bacteria in a process known as denitrification. Denitrification converts nitrogen from its compound form to its original elemental form as dinitrogen, and the cycle is ready to begin once again.