Osmosis
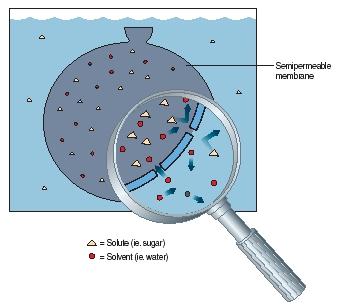
Osmosis is the movement of a solvent, such as water, through a semi-permeable membrane. (A solvent is the major component of a solution, the liquid in which something else is dissolved.) A semipermeable membrane is a material that allows some materials to flow through it but not others. The reason that semipermeable membranes have this property is that they contain very small holes. Small molecules, such as those of water, can flow easily through the holes. But large molecules, such as those of solutes (the component being dissolved, for instance sugar), cannot. Figure 1 illustrates this process. Notice that smaller molecules of water are able to pass through the openings in the membrane shown here but larger molecules of sugar are not.
Osmotic pressure
Osmosis always moves a solvent in one direction only, from a less concentrated solution to a more concentrated solution. As osmosis proceeds, pressure builds up on the side of the membrane where volume has increased. Ultimately, this pressure prevents more water from entering (for example, the bag in Figure 1), and osmosis stops. The osmotic pressure of a solution is the pressure needed to prevent osmosis from occurring.
Osmosis in living organisms
Living cells may be thought of as very small bags of solutions contained within semipermeable membranes. For example, Figure 1 might be thought of as a cell surrounded by a watery fluid. For the cell to survive, the concentration of substances within the cell must stay within a safe range.
A cell placed in a solution more concentrated than itself (a hypertonic solution) will shrink due to loss of water. It may eventually die of dehydration. You can observe this effect with a carrot placed in salty water. Within a few hours the carrot becomes limp and soft because its cells have shrivelled.
By contrast, a cell placed in a solution more dilute than itself (a hypotonic solution) will expand as water enters it. Under such conditions
the cell may burst. In general, plant cells are protected from bursting by the rigid cell wall that surrounds the cell membrane. As water enters the cell, it expands until it pushes up tight against the cell wall. The cell wall pushes back with an equal pressure, so no more water can enter.
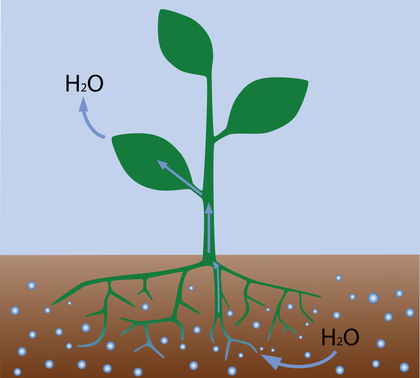
Osmosis contributes to the movement of water through plants. Solute concentrations (the ratio of solutes to solvents in a solution) increase going from soil to root cells to leaf cells. The resulting differences of osmotic pressure help to push water upward. Osmosis also controls the evaporation of water from leaves by regulating the size of the openings (stomata) in the leaves' surfaces.
Words to Know
Concentration: The quantity of solute (for example sugar) dissolved in a given volume of solution (for example water).
Hypertonic solution: A solution with a higher osmotic pressure (solute concentration) than another solution.
Hypotonic solution: A solution with a lower osmotic pressure (solute concentration) than another solution.
Isotonic solutions: Two solutions that have the same concentration of solute particles and therefore the same osmotic pressure.
Osmotic pressure: The pressure which, applied to a solution in contact with pure solvent through a semipermeable membrane, will prevent osmosis from occurring.
Semipermeable membrane: A thin barrier between two solutions that permits only certain components of the solutions, usually the solvent, to pass through.
Solute: A substance dissolved to make a solution, for example sugar in sugar water.
Solution: A mixture of two or more substances that appears to be uniform throughout except on a molecular level.
Solvent: The major component of a solution or the liquid in which some other component is dissolved, for example water in sugar water.
Organisms have various other methods for keeping their solute levels within safe range. Some cells live only in surroundings that are isotonic (have the same solute concentration as their own cells). For example, jellyfish that live in salt water have much higher salt-to-water solute concentrations than do freshwater creatures. Other animals continually replace lost water and solutes by drinking and eating. They remove excess water and solutes through excretion of urine.
Applications of osmosis
Preserving food. For thousands of years, perishable foods such as fish, olives, and vegetables have been preserved in salt or brine. The high salt concentration is hypertonic to bacteria cells, and kills them by dehydration before they can cause the food to spoil. Preserving fruit in sugar (as in jams or jellies) works on the same principle.
Artificial kidneys. People with kidney disease rely upon artificial kidney machines to remove waste products from their blood. Such machines use a process called dialysis, which is similar to osmosis. The difference between osmosis and dialysis is that a dialyzing membrane permits not just water, but also salts and other small molecules dissolved in the blood, to pass through. These materials move out of blood into a surrounding tank of distilled water. Red blood cells are too large to pass through the dialyzing membrane, so they return to the patient's body.
Desalination by reverse osmosis. Oceans hold about 97 percent of Earth's water supply, but their high salt content makes them unusable for drinking or agriculture. Salt can be removed by placing seawater in contact with a semipermeable membrane, then subjecting it to great pressure. Under these conditions, reverse osmosis occurs, by which pressure is used to push water from a more concentrated solution to a less concentrated solution. The process is just the reverse of the normal process of osmosis. In desalination, reverse osmosis is used to push water mole-cules out of seawater into a reservoir of pure water.