Genetic Engineering - Real-life applications

Big Business in DNA
Ever since the breakthrough discoveries of Watson, Crick, and others in the 1950s made genetic engineering a possibility, the new field has promised increasingly bigger payoffs. These payoffs take the form of improvements to human life and profits to those who facilitate those improvements. The possible applications of genetic engineering are virtually limitless—as are the profits to be made from genetic engineering as a business. As early as the 1970s, entrepreneurs (independent businesspeople) recognized the commercial potential of genetically engineered products, which promised to revolutionize life, technology, and commerce as computers also were doing. Thus was born one of the great buzzwords of the late twentieth century: biotechnology, or the use of genetic engineering for commercial purposes.
Several early biotechnology firms were founded by scientists involved in fundamental research: Boyer, for example, teamed up with the venture capitalist Robert Swanson in 1976 to form Genentech (Genetic Engineering Technology). Other pioneering companies, including Cetus, Biogen, and Genex, likewise were founded through the collaboration of scientists and businesspeople. Today biotechnology promises a revolution in numerous areas, such as agriculture. Recombinant DNA techniques enable scientists to produce plants that are resistant to freezing temperatures, that will take longer to ripen, that will develop their own resistance to pests, and so on. By 1988 scientists had tested more than two dozen kinds of plants engineered to have special properties such as these. Yet no field of biotechnology and genetic engineering is as significant as the applications to health and the cures for diseases.
MEDICINES AND CURES.
The use of rDNA allows scientists to produce many products that were previously available only in limited quantities: for example, insulin, which we referred to earlier. Until the 1980s the only source of insulin for people with diabetes came from animals slaughtered for meat and other purposes. The supply was never high enough to meet demand, and this drove up prices. Then, in 1982, the U.S. Food and Drug Administration (FDA) approved the sale of insulin produced by genetically altered organisms—the first such product to become available. Since 1982 several additional products, such as human growth hormone, have been made with rDNA techniques.
One of the most exciting potential applications of genetic engineering is the treatment of genetic disorders, which are discussed in Heredity, through the use of gene therapy. Among the more than 3,000 such disorders, quite a few of which are quite serious or even fatal, many are the result of relatively minor errors in DNA sequencing. Genetic engineering offers the potential to provide individuals with correct copies of a gene, which could make possible a cure for that condition. In the 1980s scientists began clinical trials of a procedure known as human gene therapy to replace defective genes. The technique, still very much in the developmental stage, offers the hope of cures for diseases that medicine has long been powerless to combat.
In 2001 scientists at the Weizmann Institute in Israel brought together two of the most exciting fields of research, biotechnology and computers, to produce the DNA-processing nanocomputer. It is an actual computer, but it is so small that a trillion of them would fit in a test tube. It consists of DNA and DNA-processing enzymes, both dissolved in liquid; thus its input, output, and software are all in the form of DNA molecules. The purpose of the nanocomputer is to analyze DNA, detecting abnormalities in the human body and creating remedies for them.
The Human Genome Project
At the center of genetic studies, with vast potential applications to genetic engineering, is the Human Genome Project (HGP), an international effort to analyze and map the DNA of humans and several other organisms. As discussed in the essay Genetics, the HGP began with efforts by the Atomic Energy Commission, a predecessor to the U.S. Department of Energy, to study the genetic effects of radioactive nuclear fallout. In 1990 the Department of Energy in cooperation with the National Institutes of Health (NIH), launched the project. At about the same time, the governments of the United Kingdom, Japan, Russia, France, and Italy initiated their own, similar undertakings, which are coordinated with American efforts.
The purpose of the project is to locate each human gene and determine its specific structure and function. Such knowledge will provide the framework for studies in health, disease, biology, and medicine during the twenty-first century and no doubt will make possible the cures for countless diseases. Although great strides have been made in gene therapy in a relatively short time, its potential usefulness has been limited by lack of scientific data concerning the multitude of functions that genes control in the human body. For gene therapy to advance to its full potential, scientists must discover the biological role of each of these genes and locate each base pair of which they are comprised.
A PROGRESS REPORT.
Scientists participating in the project have identified an average of one new gene a day, and this rate of discovery has increased. At the time of its establishment in 1990 (under the leadership of James D. Watson, who served as director until 1992), HGP was expected to reach completion by 2005. In 2002, however, the project's leadership predicted completion by some time in the following year. Along the way, they had discovered that the human genome, originally believed to include 100,000 to as many as 150,000 genes, actually consists of about 30,000 to 40,000 genes.
Both HGP and a private firm, Celera Genomics (founded 1998), had undertaken the study of the human genome, and in June 2000 the entities jointly reported that they had finished the initial sequencing of the three billion-odd base pairs in the human genome. By that point, researchers also had completed thorough DNA sequences for many other organisms. The basis for the latter undertaking is that humans share many genes with other life-forms. With the completion of initial sequencing, scientists working on the HGP undertook the effort of determining the exact sequence of the base pairs that make them up all human genes. Long before completion, the project had yielded some information. Some of the genes identified through the HGP include one that guides reproduction of the human immunodeficiency virus (HIV), which causes the acquired immunodeficiency syndrome, better known as AIDS (see Infectious Diseases). Researchers also have located a gene that predisposes people to obesity as well as genes associated with such inherited disorders as Huntington disease, Lou Gehrig disease (also called amyotrophic lateral sclerosis, or ALS), and some colon and breast cancers.
A World of Controversy
The HGP has numerous implications—and not all of them, in the minds of some critics, are positive. The NIH inadvertently created cause for concern when, in 1991, it attempted to patent certain forms of DNA. While patenting is touted as a necessary financial incentive for research initiatives, critics maintain that it restricts access to the information generated and to the use of those discoveries. That places a great deal of control in the hands of the private firm that funded the research and may limit the spread of real benefits that result from discovery.
Some scientists and politicians have raised the concern that the ability to produce detailed genetic information on people could give too much power to the people who possess that knowledge. Most states do not have laws protecting citizens against the misuse of genetic information, for instance, by employers and insurers. In the absence of effective legal remedies, genetic testing may be used to bar people from employment or insurance coverage. Insurers may even make mandatory testing a requirement for coverage. Existing laws may not be adequate to protect people's privacy: whereas the individual may be protected from having to provide potentially damaging genetic information, such information still can be obtained by testing the individual's relatives.
NIGHTMARISH IMAGES.
The idea of genetic information being used to control a person's destiny calls to mind all sorts of nightmarish images, such as those raised by the movie Gattaca (1997). Set in a dystopian, or anti-utopian, future, the film depicts a character who employs elaborate means to conceal his true identity in order to hold on to a job that otherwise would be forbidden to him because of his DNA. In fact, one does not have to go to fiction
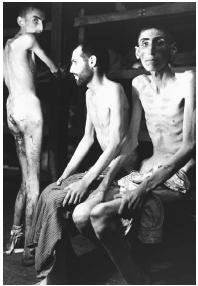
The most frightening example of this was Nazi Germany, which practiced mass murder not only of Jews and other ethnic and social groups but also of people who suffered from mental retardation or other "undesirable" traits. The purpose of DNA was discovered only a year before the collapse of the Nazi empire in 1945, and one can only imagine what Adolf Hitler and his minions would have done with this knowledge if they had had access to it. (DNA is one of several scientific and technological concepts that came to fruition at the end of World War II but which Hitler, fortunately, was unable to use to his advantage. Others include rocketry, nuclear weaponry, radar, computers, and television.)
Nazism was actually an especially repugnant version of a movement known as eugenics, which had its origins in the late nineteenth and early twentieth centuries. Based on the idea that populations could be improved by encouraging people with "positive" traits to reproduce while discouraging

OTHER CONCERNS.
The specter of eugenics raises the threat that a single human, or a group of humans, could "play God" with the lives of others. Another dramatic fear associated with genetic engineering is the threat that a genetically re-engineered virus could turn out to be extremely virulent, or deadly, and spread. There are other, more mundane questions of ethics: for instance, is it appropriate for scientists to establish private, for-profit corporations to benefit from discoveries they made while working for public-sponsored research institutions? No wonder, then, that the budget for the HGP in the United States includes a small allocation (3% of its total) toward study of the ethical, legal, and social implications (ELSI) of the project. The ELSI Working Group is charged with studying the issues of fairness, privacy, delivery of health care, and education. Meanwhile, there is a vast body of opposition to genetic engineering, biotechnology, and the HGP. And no aspect of the larger subject is more upsetting to certain individuals, as well as special interest groups, as that of cloning.
Cloning
A clone is a cell, group of cells, or organism that contains genetic information identical to that of the parent cell or organism. It is a form of asexual reproduction (see Reproduction), and as such it is not as new as it seems; what is new, however, is humans' ability to manipulate cloning at the genetic level. The first clones produced by humans as long as 2,000 years ago were plants developed from grafts and stem cuttings. By cloning—a process that calls into play complex laboratory techniques and the use of DNA replication—people usually mean a relatively recent scientific advance. Among these techniques is the ability to isolate and copy (that is, to clone) individual genes that direct an organism's development.
THE PROMISE OF CLONING.
The cloning of specific genes can provide large numbers of copies of that gene for use in genetic and taxonomic research as well as in the practical areas of medicine and farming. In the latter field, the goal is to clone plants with specific traits that make them superior to naturally occurring organisms. For example, in 1985 scientists conducted field tests using clones of plants whose genes had been altered in the laboratory to generate resistance to insects, viruses, and bacteria. New strains of plants resulting from cloning could produce crops that can grow in poor soil or even underwater and fruits and vegetables with improved nutritional qualities and longer shelf lives. A cloning technique known as twinning could induce livestock to give birth to twins or even triplets, and on the environmental front cloning might help save endangered species from extinction.
In the realm of medicine and health, cloning has been used to make vaccines and hormones. It has become possible, by combining two different kinds of cells (such as mouse and human cancer cells), to produce large quantities of specific antibodies, via the immune system, to fight off disease. When injected into the bloodstream, these cloned antibodies seek out and attack disease-causing cells anywhere in the body. By attaching a tracer element to the cloned antibodies, scientists can locate hidden cancers, and by attaching specific cancer-fighting drugs, the treatment dose can be transported directly to the cancer cells.
EXPERIMENTS IN CLONING.
The modern era of laboratory cloning began in 1958 when the British plant physiologist F. C. Steward (1904-1993) cloned carrot plants from mature single cells placed in a nutrient culture containing hormones. The first cloning of animal cells took place in 1964, when the British molecular biologist John B. Gurdon (1933-1989) took nuclei from intestinal cells of toad tadpoles and injected them into unfertilized eggs. The cell nuclei in the eggs had been destroyed with ultra-violet light, but when the eggs were incubated, Gurdon found that 1-2% of the eggs developed into fertile, adult toads.
The first successful cloning of mammals occurred nearly 20 years later, when scientists in Switzerland and the United States successfully cloned mice using a method similar to Gurdon's approach. Their method required one extra step, however: after taking the nuclei from the embryos of one type of mouse, they transferred them into the embryos of another type of mouse. The latter served as a surrogate, or replacement, mother. The cloning of cattle livestock was tried first in 1988, when embryos from prize cows were transplanted to unfertilized cow eggs whose own nuclei had been removed. An even greater breakthrough transpired on February 24, 1997, with the birth of a lamb named Dolly in Edinburgh, Scotland. Dolly was no ordinary sheep: she was the first mammal born from the cloning of an adult cell. Thus, she had been produced by asexual reproduction in the form of genetically engineered cloning rather than by anything resembling a normal process. Nonetheless, she proved her own ability to reproduce the old-fashioned way when, on April 23, 1998, she gave birth to a daughter named Bonnie.
ARE HUMANS NEXT?
Though Dolly's and Bonnie's births excited hopes, they also inspired fears. If large mammals could be cloned, could humans? As early as 1993 an attempt had been made at cloning human embryos as part of studies on in vitro (out of the body) fertilization. The purpose was to develop fertilized eggs in test tubes and then to implant them into the wombs of women having difficulty becoming pregnant. These fertilized eggs, however, did not develop to a stage that was suitable for transplantation into a human uterus. Then, on October 13, 2001, scientists at Advanced Cell Technology in Worcester, Massachusetts, successfully cloned a human embryo. They had not created human life, as it might sound; what they had developed instead was a source for nerve and other tissues that could be harvested for use in medicine and research. Still, the news—overshadowed though it was in America, where people were still reeling from the September 11 terrorist attacks—was earth-shattering. Human cells had been reproduced, and once again it appeared that the production of human clones might be possible.
It is easy to understand how people might respond with alarm to such frightening news with alarm. Such fears have a great deal more to do with Hollywood than they do with science. In fact, the accomplishment of the Massachusetts firm, while impressive from a scientific standpoint, was fairly modest compared with the Frankenstein-like image presented by anti-genetic engineering scaremongers. "Cloned an embryo" actually sounds a great deal more dramatic than what the Massachusetts scientists achieved, with just one embryo reaching the size of six cells before the cells stopped dividing. This is hardly the beginnings of a clone army.
At any rate, the cloning practiced at the Massachusetts firm was therapeutic cloning, involving the production of genetic material for the treatment of specific conditions. It is a far cry from reproductive cloning, which entails implanting a cloned embryo in a uterus—and even that is still a long way from the clichéd image of clones produced in a test tube without any parents other than the biological material used to create them.
Such ideas are related much more closely to those highlighted in Aldous Huxley's 1932 novel Brave New World than they are to scientific realities. And even if humans wanted to develop such technology, it would be many, many years in the future. As for "creating life," to do so is probably not even possible; if it is, such an achievement is about as far off as travel to another solar system. This is not to say that all fears of cloning and genetic engineering are unwarranted; on the contrary, it is good to have a healthy level of skepticism. But it is also good to be an equal-opportunity skeptic and therefore to question ideas in the popular culture—including opposition to genetic engineering.
WHERE TO LEARN MORE
Barash, David P. Revolutionary Biology: The New, Gene-Centered View of Life. New Brunswick, NJ: Transaction Publishers, 2001.
Chadwick, Ruth F. The Concise Encyclopedia of the Ethics of New Technologies. San Diego: Academic Press, 2001.
"Cloning." New Scientist (Web site). <http://www.newscientist.com/hottopics/cloning/> .
Department of Energy Human Genome Program (Web site). <http://www.ornl.gov/hgmis/> .
Genetic Engineering and Cloning: Improving Nature or Uncorking the Genie? (Web site). <http://library.thinkquest.org/19697/> .
Genetics Education Center, University of Kansas Medical Center (Web site). <http://www.kumc.edu/gec/> .
Hyde, Margaret O., and John F. Setaro. Medicine's Brave New World: Bioengineering and the New Genetics. Brookfield, CT: Twenty-First Century Books, 2001.
Judson, Karen. Genetic Engineering: Debating the Benefits and Concerns. Berkeley Heights, NJ: Enslow Publishers, 2001.
National Human Genome Research Institute (Web site). <http://www.nhgri.nih.gov> .
Twenty Facts About the Human Genome. Wellcome Trust (Web site). <http://www.wellcome.ac.uk/en/genome/thgfac.htm> .
Wade, Nicholas. Life Script: How the Human Genome Discoveries Will Transform Medicine and Enhance Your Health. New York: Simon & Schuster, 2001.
I can't get proper color change in cellulolytic enzymes assay test (DNS assay method)
please help to my project