Chapter 3
Genetic Engineering

Genes contain coded information that leads to the production of proteins. Proteins, in turn, are responsible for creating the traits that characterize individual organisms. Therefore, if a way could be found to transfer genes from one organism to another, creatures could be manufactured with traits that they had never before exhibited. Based on the description of the structure of DNA provided by Watson and Crick, researchers began to search for a way to cut genes from the DNA of one organism and paste them into another. By the 1970s, they had the answer, and the science of genetic engineering was born. It was a giant step forward. Now, a mere thirty years later, it is possible to exchange genes between one plant and another and one animal and another. It is even possible to transpose genes between plants and animals. No organism—from primitive life-forms, like bacteria, to higher order animals, like human beings—is exempt from this genetic swap meet. Genetic engineering has led to monumental advances in medicine and agriculture, but it has also given rise to a storm of controversy and debate over the limits on humankind's intrusion into the natural order of things.
Restriction Enzymes and Plasmids
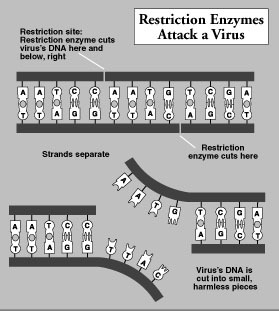
The first major breakthrough on the road to genetic engineering came with work done on restriction endonucleases by Herbert Boyer of the University of California at San Francisco. As defined by Karl Drlica in Understanding DNA and Gene Cloning: A Guide for the Curious , restriction endonucleases "are a group of enzymes [a special type of protein] that . . . occur naturally in a large number of different bacterial species, serving as part of the natural defense mechanism that protects bacterial cells against invasion by foreign DNA molecules such as those contained in viruses." 15
When, for example, a virus attacks a single-celled bacterium, restriction endonucleases are unleashed and go to work, cutting the invading DNA into small, nonthreatening pieces. "Crucial to this protective device is the ability of the nuclease to discriminate between its own DNA and the invading DNA; otherwise the cell would destroy its own DNA," Drlica says.
This recognition process involves two elements. First there are specific nucleotide sequences [As and Ts, Cs and Gs] that act as targets for the nuclease. These are called the restriction sites. Second, there is a protective chemical signal that can be placed by the cell on all the target sequences that happen to occur in its own DNA. The signal modifies the DNA and prevents the nuclease from cutting. Invading DNAs, lacking the protective signal, would be chopped by the nuclease. 16
Thus, restriction enzymes have the remarkable ability to recognize specific arrangements of DNA base pairs—As and Ts, Gs and Cs. They also have the capacity to act like a molecular scalpel, severing the DNA at exactly the spot where they detect this sequence of genetic letters. Restriction enzymes are a powerful tool because there are thousands of them, and each one acts only on a unique arrangement of As and Ts and Cs and Gs.
A second piece of the genetic engineering puzzle fell into place when it was discovered that bacteria
Recombinant DNA Technology
Boyer and Stanley Cohen, another scientist at the University of California
who was working on plasmids, pooled their knowledge to conduct a series of
experiments on two different strains of the E. coli bacteria. Some forms
of E. coli live in the intestines of humans and other animals, where they
aid the body's digestive processes. Boyer and Cohen marshaled
restriction endonucleases to cut some E. coli plasmids. When plasmids are
cut, they leave what researchers call "sticky ends," to
which other plasmid segments can easily attach themselves. The point at
which the pieces of the two plasmids join is cemented by the activity of
an enzyme called ligase, which can be described as molecular glue, to form
a stable chemical bond. Then, the two scientists severed particular genes
from another type of bacteria, one that was resistant to antibiotics, and
spliced them to the sticky ends of the cut E. coli plasmids. The result: a
hybrid form of antibiotic-resistant
E. coli.
One big question remained to be answered. Thus far,

E. coli bacterial cell. When the bacterial cell started to multiply, the scientists analyzed each successive generation and found that they all contained the tadpole gene. The first gene transfer between species had been accomplished, and the door was now open to a wide range of similar experiments—many of them far more controversial. It had been practically demonstrated that genes from fish, even genes from plants, could be transplanted into humans.
The new technique was called recombinant DNA technology—just another name for genetic engineering—because the procedure recombined genes that originated in different organisms. The popular media gave it another name that has been responsible for a great deal of confusion. They called it gene cloning, creating the belief that science could duplicate entire organisms, an achievement that was not at that point even distantly attainable. But what the technique did allow scientists to do was create specific types of proteins in large quantities. "Usually, a specific protein is produced by a cell in very small quantities, sometimes a mere one or two molecules per cell," says Frank-Kamenetskii.
As a result, the production of proteins needed for particular research was an arduous and costly undertaking. One had to process dozens of kilograms [a unit of weight equal to 2.2 pounds], nay tons, of biomass to obtain milligrams of protein.
Despite such meager quantities, it was still not possible to ensure the necessary purity of the protein. Hence, the cost of many protein preparations was exorbitant and their purity was substandard. Genetic engineering brought about a radical change in this situation. Genetic-engineering strains now exist—superproducers of many proteins with high standards of purity—that were undreamed of before. Molecular biology firms have sharply diversified the production of enzymes and other protein preparations and have reduced the prices of these products. Thus, molecular biology received a powerful new impetus, resulting in an unheard of acceleration in the pace of scientific research. 18
In accomplishing this goal, bacteria, especially E. coli bacteria, have proved to be the most effective host for transplanted genes because they reproduce rapidly. For example, if scientists wish to produce a certain kind of protein, they snip the required genes from an animal that produces the protein naturally and transplant them into an E. coli cell. They then put the cell in an environment that encourages it to divide and just let nature take its course until they have millions of cells all producing the desired protein. Finally, the scientists extract the protein from the cells and use it for whatever purpose they have in mind.
Biofoods
The first area in which the new science of genetic engineering took hold was agriculture. It quickly became apparent that food plants could be genetically altered so they were more resistant to pests, needed less water to grow, and provided more nutrition than in their natural states. Since human beings first began to till the land, farmers have been trying to produce hardier, more profitable crops. The method

Genetic engineering takes the guesswork out of this effort and greatly reduces the time it takes to produce a plant with the desired traits. It also—for the first time—makes it possible to breed entirely different types or species of plants with each other to create some truly novel hybrids. Previously, selective breeding limited farmers to experiments with plants of the same or very closely related species. By cutting and pasting genes from one plant to another, genetic engineers are able to do all the things that crossbreeding can do, and do them faster and more accurately.
The technique also allows scientists to do things that nature alone is incapable of doing. A report prepared at University of Virginia on the state of currently available genetically engineered, or transgenic, plants states:
Many plants have been commercialized, including tomatoes and squash and commodity crops like corn and soybeans. Most have been engineered for one of three traits: herbicide [weed killer] tolerance, insect resistance, or virus tolerance. This is the fastest growing area of biotechnology in agriculture. Genetically engineered cotton has been approved for commercial use. There are between 10 and 12 million acres of cotton in the U.S. and estimates are that all of this acreage will be planted to transgenic varieties within the next 10 years. One of the newest innovations in cotton is the development of naturally-colored cotton fibers where the pigments have come from inserting color genes from flowers into cotton. 19
As a further indication of the widespread use of genetic engineering in farming, the Food and Agriculture Organization of the United Nations, citing figures for the year 1999 (the process of gathering such information accurately is slow when developing countries are involved), reports:
Transgenic plants . . . now cover large areas in certain parts of the world. Estimates for 1999 indicate that 39.9 million hectares [a unit of measurement equivalent to 2.47 acres] were planted with transgenic crops. . . . Of the 39.9 million hectares, 28.1 million (i.e. 71%) were modified for tolerance to a specific herbicide (which could be sprayed on the field, killing weeds while leaving the crop undamaged); 8.9 million hectares (22%) were modified to include a toxin-producing gene from a soil bacterium . . . which poisons insects feeding on the plants, while 2.9 million hectares (7%) were planted with crops having both herbicide tolerance and insect resistance. 20
The bioengineering of plants has become big business. Hundreds of millions of dollars of research money are being poured into a diverse range of projects. Among the most promising are creating plants that produce their own fertilizer and modifying plants to be delivery systems for medicines and nutrients they do not naturally produce. For example, work is under way to produce a banana that contains in its DNA a vaccine for hepatitis B, a highly contagious disease that damages the livers of people who contract it. The banana is also being turned into a megavitamin to deliver much-needed nutrients to children in the underdeveloped countries of Africa and Asia.
Transgenic Animals
Plants are not the only organisms that genetic engineers are working on. Turning their attention to animals, scientists have produced a number of transgenic creatures they hope will bring major benefits to mankind. For example, human genes have been put into pigs to allow the pigs to produce human insulin, a substance needed to control diabetes, one of the fastest-growing diseases in affluent countries.
The applications are wide-ranging. Goats are being genetically modified to produce a protein that aids in blood clotting. Other experiments with goats aim to find cures for multiple sclerosis and some forms of cancer. Sheep are being altered to generate a protein that may fight the lung disease emphysema. Designer dairy products are also on the drawing boards. Geneticists hope to end up with a breed of cow that produces, for example, only low-fat milk.
Other genetically engineered animals are being designed to contract human diseases so that experimental treatments can be explored. In these cases, healthy genes are replaced with malfunctioning counterparts, using a technique similar to the cut-and-paste procedure used with plasmids.
Finally, researchers are optimistic that they will be able to turn animals, principally pigs, into sources of organs for human transplants. To accomplish this, they are transferring human genes into pigs so that the resulting organs will more closely resemble those found in humans and thus be less likely to be rejected.
Cloning
Work on transgenic animals has also led to the cloning of entire organisms. A clone is an identical genetic copy of an organism—its DNA is the same as that of the original from which the copy was made. In humans, identical twins are naturally occurring clones. In these cases, the original fertilized egg divides into two genetically identical halves and proceeds to develop into two distinct babies. Since the babies originated from the same egg fertilized by the same sperm, they have exactly the same DNA. By contrast, fraternal twins come from two separate eggs, each of which is fertilized by a different sperm cell. Even though these children are born at the same time, they are as genetically different from each other as any other pair of siblings born years apart would be.
The cloning of organisms must be carefully distinguished from the cloning of genes—a distinction that the popular media have not always succeeded in making. The cloning of single genes, using plasmids, is an established procedure; the cloning of organisms is still experimental and highly controversial. Although several fringe groups claim to have successfully cloned human beings, they have failed—as of the writing of this book—to produce any evidence to support their contentions.
To this date, the most famous cloned mammal remains Dolly the sheep. A close look at how Dolly was created will provide a good description of the techniques required to clone any higher animal, including humans. In announcing Dolly's birth in 1997, Scientific American magazine reported:
Dolly, unlike any other mammal that has ever lived, is an identical copy of another adult and has no father. She is a clone, the creation of a group of veterinary researchers. That work, performed by Ian Wilmut and his colleagues at the Roslin Institute in Edinburgh, Scotland, has provided an important new research tool and has shattered a belief widespread among biologists that cells from adult mammals cannot be persuaded to regenerate a whole animal. 21
Previously, researchers had cloned mammals and other animals using embryonic cells as a starting point. Embryonic cells, taken from an undeveloped fertilized egg, are different from adult cells in that they are undifferentiated. When an egg cell is fertilized, it starts to divide. Up to a certain point, the cells in each succeeding generation have the ability to develop into specialized cells that will make up the various parts (organs, bones, skin, etc.) of the mature organism. After that point, cells become differentiated, or specialized—some of them begin to turn into liver cells, others into brain cells, and so on. Until Dolly, it was thought that clones could be produced only from undifferentiated cells that would divide and grow to maturity as the cloned organism developed.
Promise and Problems of Cloning
Dolly, however, was cloned from a cell taken from the udder of a six-year-old female sheep, a fully developed adult. Dolly was the 277th attempt made by Wilmut and his fellow researchers. The other attempts had

Once the transfer was complete, the recipient eggs contained a complete set of genes, just as they would do if they had been fertilized by sperm. The eggs were then cultured for a period before being implanted into sheep that carried them to term, one of which culminated in a successful birth. The resulting lamb was, as expected, an exact genetic copy, or clone, of the sheep that provided the transferred nucleus, not of [the sheep] that provided the egg. 22
Wilmut used a pipette many times thinner than a human hair to remove the DNA from the host egg. Then the empty egg was placed next to a cell taken from the donor sheep's udder and the two were fused together using a tiny jolt of electricity. Another pulse of electricity caused the egg cell, with its new DNA, to start dividing. The cell was now behaving just like a normal egg cell would if it had been fertilized by sperm from a male sheep. It was cultured for a few days in a laboratory dish and then implanted into the uterus of a third sheep, which carried it to term and gave birth to Dolly.
From the beginning, Wilmut and other geneticists were concerned that since Dolly's DNA came from a six-year-old sheep she might age prematurely. At first, she seemed to be perfectly healthy and gave birth to a lamb of her own in 1998. But then Dolly began to develop medical problems frequently associated with aging. "Early in life Dolly had a weight problem. Then in 1999, it emerged that caps at her chromosome ends called telomeres, which get shorter each time a cell divides, were 20 percent shorter than

In 2002, Dolly was diagnosed with arthritis, another disease associated with old age, and then she came down with lung cancer. Dolly was humanely put to death at the age of six and a half years, half the normal life span of a sheep of her kind. Cloned animals since Dolly, including cows, rabbits, mice, cats, goats, and pigs, have experienced similar problems. "Dolly's premature death is typical of cloned animals," Whitfield writes.
From conception onwards, clones suffer a higher mortality rate than non-clones. Studies in mice seem to show that this bad health persists throughout life. Some seized upon Dolly's ailments as evidence that clones are invariably sickly and age prematurely. Although it can't be ruled out that her origins made her less robust than other sheep, it is not possible to make generalizations about clones' health from the fate of a single animal. . . . But the process of genetic reprogramming seems too complex and haphazard to control tightly, and its success rate has not improved much since Dolly's day. 24
Although many questions about animal cloning remain to be answered, scientists are hopeful that, as

The Human Genome Project
The human genome is composed of about 3 billion base pairs of As and Ts, Gs and Cs. In 1990, an international consortium of scientists set out to create a map that would show exactly where on our twenty-three pairs of chromosomes every one of those base pairs is located. The effort, called the Human Genome Project, is the most extensive scientific enterprise ever undertaken.
Genetic mapping is the first step in isolating a gene. There are two basic approaches to this complex endeavor. The first is called linkage mapping, and its goal is to show where on each chromosome each gene is located relative to other genes. The method is to compare the genetic makeup of members of a family or closely related group of people who have a history of a specific disease. By finding similar base pairs on the chromosomes of family members in succeeding generations of this group who have the disease, scientists can isolate the responsible gene and begin to build a map of the entire genome. The second type of map is far more ambitious. It is called a physical map and its goal is to show the absolute (not relative) location of the base pairs that make up every gene.
In both cases, the cut-and-paste techniques developed by genetic engineering are indispensable. Restriction enzymes are used to cut the chromosomes into small segments, which are then cut into even smaller pieces until the sought-after gene is found. It is a painstaking process, made possible not only by the methods of genetic engineering, but also by powerful supercomputers that enable researchers to compare various overlapping segments to weed out duplicate base pairs and base pairs that appear not to play any role in the process of genetic inheritance. Only about 2 percent of the 3 billion base pairs actually make up functional genes. The rest, called junk DNA, help to locate the genes and may play other roles that remain to be determined.
The Human Genome Project was virtually completed in the year 2003. It yielded many interesting—and surprising—insights into the genetic composition of human beings. Among them: the total number of genes in a human being lies between 30,000 and 35,000, far fewer than earlier estimates of 80,000 to 140,000; the average gene consists of about 3,000 base pairs, but sizes vary greatly, the longest being 2.4 million base pairs; 99.9 percent of base pairs are identical in all people; the function of more than 50 percent of human genes has not yet been determined. While work continues on tracking down the roles played by these mystery genes, the next step is the mapping of the human proteome—the complete array of proteins operating in the human body. This includes pinpointing all the proteins coded for by the genes and describing the specific role these proteins play in the human organism.
PLEASE EMAIL THANKS!
thanks
This provides us the best information on Genetics and molecular biology