Carbon cycle

The carbon cycle is the process in which carbon atoms are recycled over and over again on Earth. Carbon recycling takes place within Earth's biosphere and between living things and the nonliving environment. Since a continual supply of carbon is essential for all living organisms, the carbon cycle is the name given to the different processes that move carbon from one to another. The complete cycle is made up of "sources" that put carbon back into the environment and "sinks" that absorb and store carbon.
Recycling carbon
Earth's biosphere can be thought of as a sealed container into which nothing new is ever added except the energy from the Sun. Since new matter can never be created, it is essential that living things be able to reuse the existing matter again and again. For the world to work as it does, everything has to be constantly recycled. The carbon cycle is just one of several recycling processes, but it may be the most important process since carbon is known to be a basic building block of life. As the foundation atop which a huge family of chemical substances called organic substances are formed, carbon is the basis of carbohydrates, proteins, lipids, and nucleic acids—all of which form the basis of life on Earth.
Since all living things contain the element carbon, it is one of the most abundant elements on Earth. The total amount of carbon on Earth, whether we are able to measure it accurately or not, always remains the same, although the carbon regularly changes its form. A particular carbon atom located in someone's eyelash may have at one time been part of some now-extinct species, like a dinosaur. Since the dinosaur died and decomposed millions of years ago, its carbon atoms have seen many forms before ending up as part of a human being. It may have been part of several plants and trees, free-floating in the air as carbon dioxide, locked away in the shell of a sea creature and then buried at the ocean bottom, or even part of a volcanic eruption. Carbon is found in great quantities in Earth's crust, its surface waters, the atmosphere, and the mass of green plants. It is also found in many different chemical combinations, including carbon dioxide (CO 2 ) and calcium carbonate (CaCO 3 ), as well as in a huge variety of organic compounds such as hydrocarbons (like coal, petroleum, and natural gas).
Words to Know
Biosphere: The sum total of all life-forms on Earth and the interaction among those life-forms.
Decomposition: The breakdown of complex molecules—molecules of which dead organisms are composed—into simple nutrients that can be reutilized by living organisms.
Fossil fuel: A fuel such as coal, oil, or natural gas that is formed over millions of years from the remains of plants and animals.
Greenhouse effect: The warming of Earth's atmosphere due to water vapor, carbon dioxide, and other gases in the atmosphere that trap heat radiated from Earth's surface.
Hydrocarbons: Molecules composed solely of hydrogen and carbon atoms.
Photosynthesis: Chemical process by which plants containing chlorophyll use sunlight to manufacture their own food by converting carbon dioxide and water to carbohydrates, releasing oxygen as a by-product.
Respiration: The process in which oxygen is used to break down organic compounds into carbon dioxide and water.
Carbon cycle processes
If a diagram were drawn showing the different processes that move carbon from one form to another, its main processes would be photosynthesis, respiration, decomposition, natural weathering of rocks, and the combustion of fossil fuels.
Photosynthesis. Carbon exists in the atmosphere as the compound carbon dioxide. It first enters the ecological food web (the connected network of producers and consumers) when photosynthetic organisms, such as plants and certain algae, absorb carbon dioxide through tiny pores in their leaves. The plants then "fix" or capture the carbon dioxide and are able to convert it into simple sugars like glucose through the biochemical process known as photosynthesis. Plants store and use this sugar to grow and to reproduce. Thus, by their very nature as makers of their own food, plants remove carbon dioxide from the atmosphere. When plants are eaten by animals, their carbon is passed on to those animals. Since animals cannot
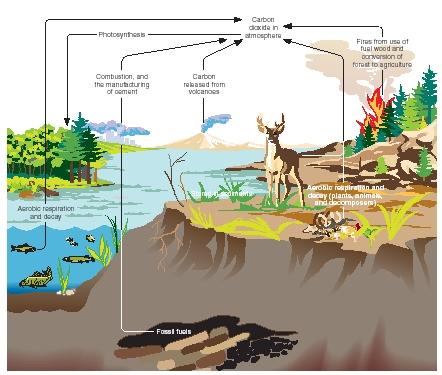
make their own food, they must get their carbon either directly by eating plants or indirectly by eating animals that have eaten plants.
Respiration. Respiration is the next step in the cycle, and unlike photosynthesis, it occurs in plants, animals, and even decomposers. Although we usually think only of breathing oxygen when we hear the word "respiration," it has a broader meaning that involves oxygen. To a biologist, respiration is the process in which oxygen is used to break down organic compounds into carbon dioxide (CO 2 ) and water (H 2 O). For an animal then, respiration is both taking in oxygen (and releasing carbon dioxide) and oxidizing its food (or burning it with oxygen) in order to release the energy the food contains. In both cases, carbon is returned to the atmosphere as carbon dioxide. Carbon atoms that started out as components of carbon dioxide molecules have passed through the body of living organisms and been returned to the atmosphere, ready to be recycled again.
Decomposition. Decomposition is the largest source through which carbon is returned to the atmosphere as carbon dioxide. Decomposers are microorganisms that live mostly in the soil but also in water, and which feed on the rotting remains of plants and animals. It is their job to consume both waste products and dead matter, during which they also return carbon dioxide to the atmosphere by respiration. Decomposers not only play a key role in the carbon cycle, but also break down, remove, and recycle what might be called nature's garbage.
Weathering of rocks. Not all carbon atoms are always moving somewhere in the carbon cycle. Often, many become trapped in limerock, a type of stone formed on the ocean floor by the shells of marine plankton. Sometimes after millions of years, the waters recede and the limerock is eventually exposed to the elements. When limerock is exposed to the natural process of weathering, it slowly releases the carbon atoms it contains, and they become an active part of the carbon cycle once again
Human-caused increase of carbon dioxide in the atmosphere. In recent history, humans have added to the carbon cycle by burning fossil fuels. Ever since the rapid growth of the Industrial Revolution in the nineteenth century when people first harnessed steam to power their engines, human beings have been burning carbon-containing fuels like coal and oil (called fossil fuels) for artificial power. This constant burning produces massive amounts of carbon dioxide, which are released into Earth's atmosphere. Over the last 150 years, the burning of coal, oil, and natural gas has released some 270 billion tons (245 billion metric tons) of carbon into the air in the form of carbon dioxide.
Luckily, more than half of the carbon dioxide emitted by the burning of fossil fuels is absorbed by the oceans, by plants, and by soils. Regardless, scientists feel fossil fuel consumption could be an example of a human activity that affects and possibly alters the natural processes (photosynthesis, respiration, decomposition) that nature had previously kept in balance. Many scientists believe that carbon dioxide is a "greenhouse gas." This means that it traps heat and prevents it from escaping from Earth. As a result, this trapped gas leads to a global temperature rise, a natural phenomenon known as the greenhouse effect, which can have disastrous effects on Earth's environment.
[ See also Greenhouse effect ]
send me some information about carbon cycle, carbon trading, Kyoto protocol and the greenhouse effect.
thanks anyways!!!
awsome description and illustartion
O_o Cindy O_o //_^
Good work!
i have photosynthesis, consumption, combustion, respiration and dissolving but i dont know what process they all belong too.
Keep it up!!
Thanks.
thank you in advance
its great and its well understood..
God bless you all.. ^^
Although I do agree with Caitlyn, You really need a better background.
Also, what else can you call the TCA cycle?
Thank you. Most helpful.
you guys are awesome!
This really helped with my Chemistry assignments
And for the average carbon atom in a tree, how long ago was that atom in the air?
Thank you!!!
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle. Carbon is found in all living things. Carbon atoms move constantly through
living organisms, the oceans, the atmosphere, and the Earth’s crust in what is
known as the carbon cycle. The directions taken by carbon atoms through this
cycle are very complicated and can take millions of years to make a full circle.